Researchers propose a new method that might be used to screen vehicles, suspicious packages, or cargo from up to a few hundred meters away
From the Journal: Physics of Plasmas
WASHINGTON, D.C., March 15, 2016 – In 2004 British national Dhiren Barot was arrested for conspiring to commit a public nuisance by the use of radioactive materials, among other charges. Authorities claimed that Barot had researched the production of “dirty bombs,” and planned to detonate them in New York City, Washington DC, and other cities. A dirty bomb combines conventional explosives with radioactive material.
Although Barot did not build the bombs, national security experts believe terrorists continue to be interested in such devices for terror plots. Now researchers from the University of Maryland have proposed a new technique to remotely detect the radioactive materials in dirty bombs or other sources. They describe the method in a paper in the journal Physics of Plasmas, from AIP Publishing.
While the explosion of a dirty bomb would likely cause more damage than the radioactive substances it spreads, the bombs could create fear and panic, contaminate property, and require potentially costly cleanup, according to the U.S. Nuclear Regulatory Commission.
Radioactive materials are routinely used at hospitals for diagnosing and treating diseases, at construction sites for inspecting welding seams, and in research facilities. Cobalt-60, for example, is used to sterilize medical equipment, produce radiation for cancer treatment, and preserve food, among many other applications. In 2013 thieves in Mexico stole a shipment of cobalt-60 pellets used in hospital radiotherapy machines, although the shipment was later recovered intact.
Cobalt-60 and many other radioactive elements emit highly energetic gamma rays when they decay. The gamma rays strip electrons from the molecules in the surrounding air, and the resulting free electrons lose energy and readily attach to oxygen molecules to create elevated levels of negatively charged oxygen ions around the radioactive materials.
It’s the increased ion density that the University of Maryland researchers aim to detect with their new method. They calculate that a low-power laser aimed near the radioactive material could free electrons from the oxygen ions. A second, high-power laser could energize the electrons and start a cascading breakdown of the air. When the breakdown process reaches a certain critical point, the high-power laser light is reflected back. The more radioactive material in the vicinity, the more quickly the critical point is reached.
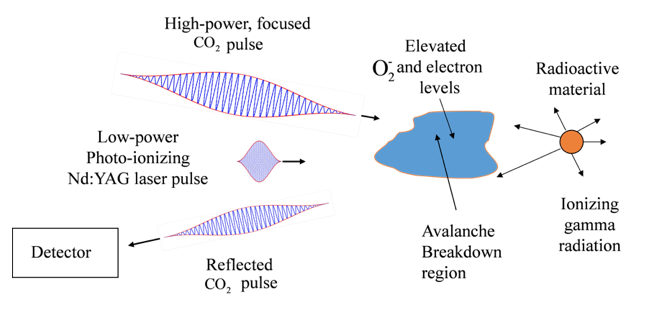
“We calculate we could easily detect 10 milligrams [of cobalt-60] with a laser aimed within half a meter from an unshielded source, which is a fraction of what might go into a dirty bomb” said Joshua Isaacs, first author on the paper and a graduate student working with University of Maryland physics and engineering professors Phillip Sprangle and Howard Milchberg. Lead could shield radioactive substances, but most ordinary materials like walls or glass do not stop gamma rays.
The lasers themselves could be located up to a few hundred meters away from the radioactive source, Isaacs said, as long as line-of-sight was maintained and the air was not too turbulent or polluted with aerosols. He estimated that the entire device, when built, could be transported by truck through city streets or past shipping containers in ports. It could also help police or security officials detect radiation without being too close to a potentially dangerous gamma ray emitter.
The proposed remote radiation detection method is not the first, but it has advantages over other approaches. For example, terahertz radiation has also been proposed as a way to breakdown air in the vicinity of radioactive materials, but producing terahertz radiation requires complicated and costly equipment. Another proposed method would use a high-power infrared laser to both strip electrons and break down the air, but the method requires the detector be located in the opposite direction of the laser, which would make it impractical to create a single, mobile device.
So far the researchers at the University of Maryland have analyzed the feasibility of the new approach and experiments are underway to test it in the lab.
Isaacs said it would be difficult to estimate when a detection device based on the new method might be commercialized, but he didn’t foresee a specific manufacturing challenge that would stand in its way.
“We specifically chose well developed technology for each component of the proposed system,” he said.
###
For More Information:
AIP Media Line
media@aip.org
301-209-3090
Article Title
Remote Monostatic Detection of Radioactive Materials by Laser-induced Breakdown
Authors
Joshua Isaacs, Chenlong Miao and Phillip Sprangle
Author Affiliations
University of Maryland and Naval Research Laboratory
