Effects of high-fat diet are more impactful than glucose, insulin, and ketone levels in triple-negative breast cancer.
From the Journal: APL Bioengineering
WASHINGTON, March 3, 2026 — If you’re diagnosed with breast cancer, what should you eat to ensure the best prognosis?
In APL Bioengineering, by AIP Publishing, a multidisciplinary team of researchers at Princeton University conducted a study to find out.
“We took the approach of building identical engineered tumors and culturing them in conditions that mimic the blood composition of patients under different dietary states,” author Celeste M. Nelson said. “We were hoping to identify dietary conditions that would slow tumor growth. Instead, we found one dietary condition — a high-fat diet — that sped up tumor growth.”
The researchers engineered a tumor model using a human plasmalike medium to re-create a more realistic microenvironment around tumors. This allowed them to replicate the biochemical effects of nutrients from food. As a result, they could isolate specific nutrients and their effects and closely examine the metabolic reprogramming that occurs in cancer cells.
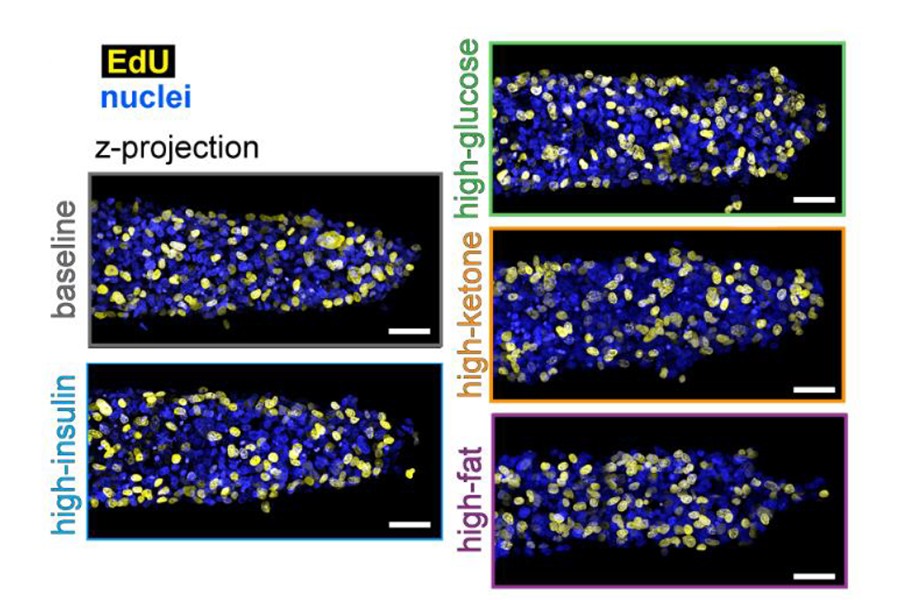
Their study focused on triple-negative breast cancer, a subtype that is particularly difficult to treat with standard methods. They carefully examined the structure, growth, and spread of cancer cells and how these characteristics differ in four different dietary conditions that can occur in a human body: high-insulin, high-glucose, high-ketone, and high-fat.
They discovered a high-fat diet accelerates tumor growth and invasion. They also found it causes an increase in the enzyme MMP1, which degrades the extracellular matrix, and is associated with a poor prognosis. Using their results, the researchers will be able to apply their method to other breast cancer subtypes and scenarios.
Previous studies to examine the connection between diet and tumor growth fall short by failing to account for the complexity of interconnected systems in the body. The interplay between the immune system, human tissues involved in metabolism, and the microbiome of trillions of microorganisms in the body affects how cancer cells behave.
In addition, cells in the body are bathed in a water-based fluid, called interstitial fluid, that flows continuously around cells. Earlier studies examining how nutrients from food affect tumors have struggled to replicate the constant flow of nutrients around cells.
“Cells are typically cultured in media that is saturated with sugars and other biochemicals at levels that don’t match what you see in the human body,” Nelson said. “Our study shows that tumor cells behave differently when cultured in media that matches the biochemical composition of human plasma.”
The researchers plan to use their results to further examine the complex interplay between dietary conditions and various tumor therapies.
“We plan to take the same system and define whether tumors respond differently to chemotherapy when cultured in media mimicking the different dietary conditions,” Nelson said. “This would allow physicians to potentially make recommendations about what a patient should eat if prescribed a specific therapy.”
###
Article Title
Fat promotes growth and invasion in a 3D microfluidic tumor model of triple-negative breast cancer
Authors
Maryam Kohram, Carolina Trenado-Yuste, Molly C. Brennan-Smith, Evelyn S. Navarro Salazar, Pengfei Zhang, Jasmine E. Hao, Xincheng Xu, Bharvi Chavre, William Oh, Sherry X. Zhang, Susan E. Leggett, Rolf-Peter Ryseck, Joshua D. Rabinowitz, and Celeste M. Nelson
Author Affiliations
Princeton University
