From the Journal: Journal of Applied Physics
WASHINGTON, D.C., September 3, 2019 — An explosion is a complex event involving quickly changing temperatures, pressures and chemical concentrations. In a paper in the Journal of Applied Physics, from AIP Publishing, a special type of infrared laser, known as a swept-wavelength external cavity quantum cascade laser (swept-ECQCL), is used to study explosions. This versatile instrument has a broad wavelength tuning range that allows the measurement of multiple chemical substances, even large molecules, in an explosive fireball.
The ability to measure and monitor the dramatic changes during explosions could help scientists understand and even control them. Measurements using rugged temperature or pressure probes placed inside an exploding fireball can provide physical data but cannot measure chemical changes that may be generated during the explosion. Sampling the end products of a detonation is possible but provides information only once the explosion is over.
In this work, molecules in the fireball are detected by monitoring the way they interact with light, especially in the infrared region. These measurements are fast and can be taken a safe distance away. Since fireballs are turbulent and full of strongly absorbing substances, lasers are needed.
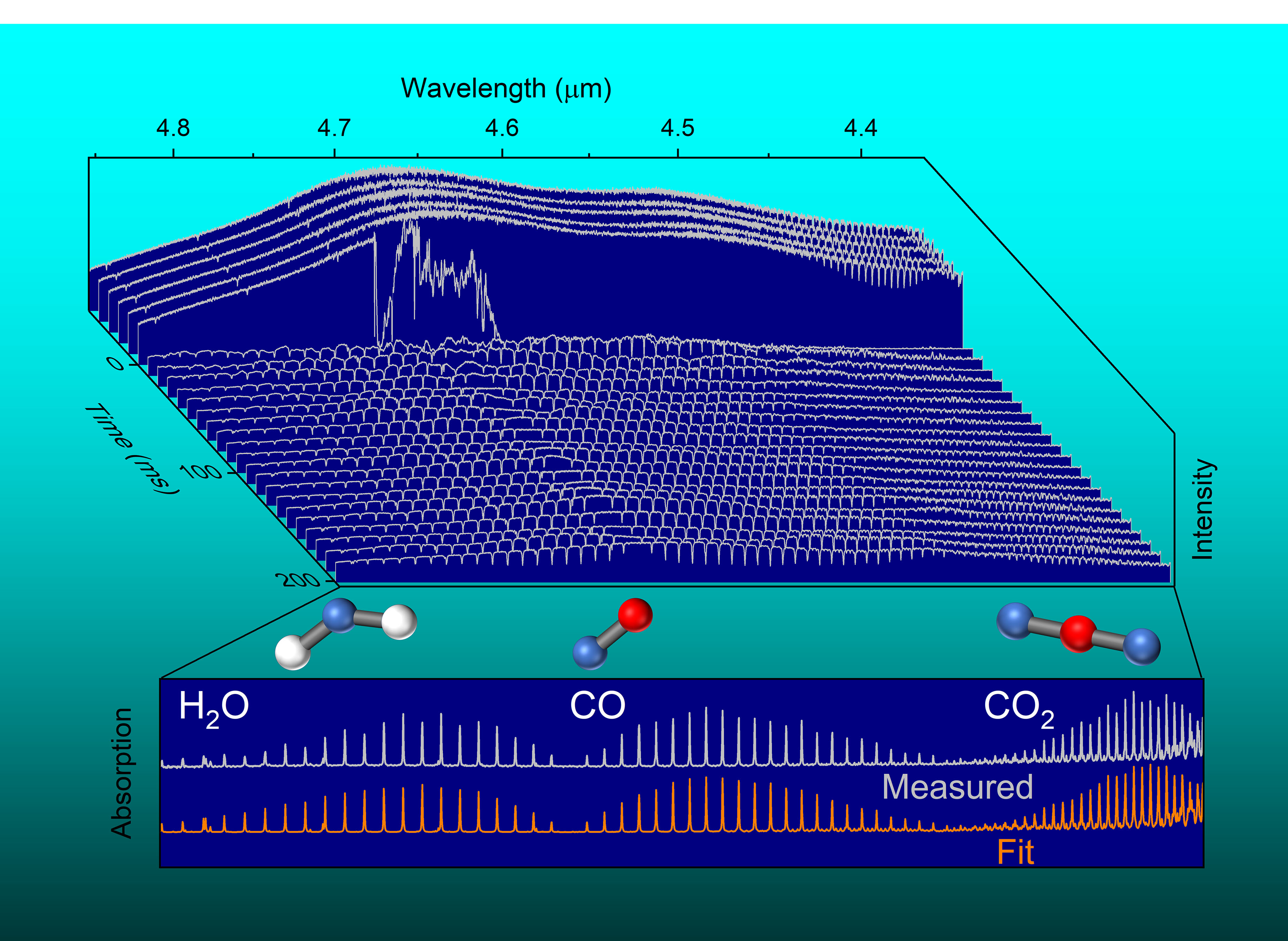
Using a new instrument built in their lab, the investigators measured explosive events at faster speeds, at higher resolutions and for longer time periods than previously possible using infrared laser light.
“The swept-ECQCL approach enables new measurements by combining the best features of high-resolution tunable laser spectroscopy with broadband methods such as FTIR,” co-author Mark Phillips explained.
The study looked at four types of high-energy explosives, all placed in a specially designed chamber to contain the fireball. A laser beam from the swept-ECQCL was directed through this chamber while rapidly varying the laser light’s wavelength. The laser light transmitted through the fireball was recorded throughout each explosion to measure changes in the way infrared light was absorbed by molecules in the fireball.
The explosion produces substances such as carbon dioxide, carbon monoxide, water vapor and nitrous oxide. These can all detected by the characteristic way each absorbs infrared light. Detailed analysis of the results provided the investigators with information about temperature and concentrations of these substances throughout the explosive event. They were also able to measure absorption and emission of infrared light from tiny solid particles (soot) created by the explosion.
The swept-ECQCL measurements provide a new way to study explosive detonations that could have other uses. In future studies, the investigators hope to extend the measurements to more wavelengths, faster scan rates, and higher resolutions.
###
For more information:
Larry Frum
media@aip.org
301-209-3090
Article Title
Authors
Mark C. Phillips, Bruce E. Bernacki, Sivanandan S. Harilal, Brian E. Brumfield, Joel M. Schwallier and Nick G. Glumac
Author Affiliations
Pacific Northwest National Laboratory, University of Arizona, Opticslah LLC and University of Illinois at Urbana-Champaign
