Using Debye lengths, researchers in Japan improve sensor response to better analyze biomolecules.
From the Journal: Journal of Applied Physics
WASHINGTON, D.C., August 16, 2018 — In clinical diagnostics, it is critical to monitor biomolecules in a simple, rapid and sensitive way. Clinicians most often monitor antibodies because these small proteins attach to antigens, or foreign substances, we face every day. Most biomolecules, however, have complicated charge characteristics, and the sensor response from conventional carbon nanotube systems can be erratic. A team in Japan recently revealed how these systems work and proposed changes to dramatically improve biomolecule detection. They report their findings in the Journal of Applied Physics, from AIP Publishing.
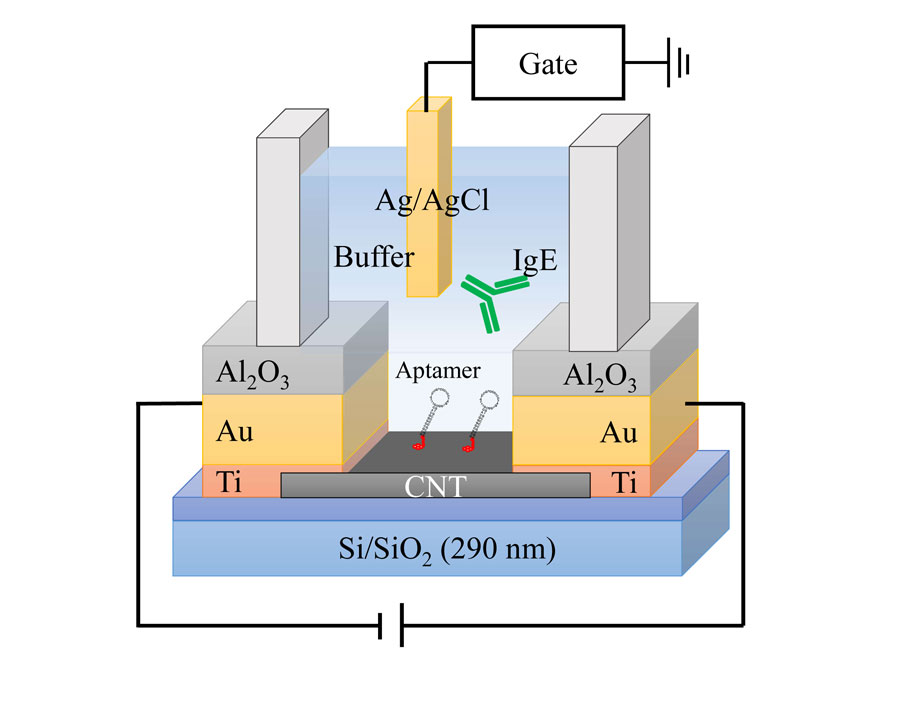
These researchers demonstrated a new technique to detect, measure and analyze biomolecules with inhomogeneous charge distributions by adjusting the solution in which they monitor the biomolecule. They used carbon nanotube thin film transistors (CNT-TFTs) to zero in on the precise amount of a specific biomolecule is in a specimen.
CNT-TFT biosensors use immune antibody receptors called aptamers to detect the net electric charge of the part of the target molecule. After scientists identify a molecule, an antibody is made to attach to it in solution. That antibody then connects to an aptamer on a thin film of carbon nanotubes that converts the connection into an electrical signal for sensor detection. With this enhanced sensor response, researchers can determine the Debye length, or the distance between a point charge and the molecule, to map out a molecule’s uneven charge distributions.
The group discovered that they had to look at how the charges were distributed close to a molecule’s surface to understand the complicated behavior in the sensor signal. “Despite being the same target molecule, the polarities of the sensor response are completely different from positive or negative,” said Ryota Negishi, an author on the paper.
“We achieved the improvement of dynamic range by using low concentration of buffer solution,” Negishi said. “As a result, we clarified the mechanism of complicated sensor response which has not been clarified in previous reports.”
Many different features of an experiment can affect a molecule’s Debye length, so these results show promise for further controlling sensors and modifying their dynamic range.
Next, Negishi and his colleagues hope to find a way to use their findings in more real-life scenarios. “For practical application, it is essential to develop a sensing technology that can be detected under high concentration conditions close to blood.”
###
For More Information:
Rhys Leahy
media@aip.org
301-209-3090
@AIPPhysicsNews
Article Title
Biosensor response from target molecules with inhomogeneous charge localization
Authors
Hiroto Kase, Ryota Negishi, Michiharu Arifuku, Noriko Kiyoyanagi and Yoshiro Kobayashi
Author Affiliations
Osaka University and Nippon Kayaku Company, Ltd.
