From the Journal: Physics of Plasmas
WASHINGTON, D.C., August 23, 2019 — When energy is added to uranium under pressure, it creates a shock wave, and even a tiny sample will be vaporized like a small explosion. By using smaller, controlled explosions, physicists can test on a microscale in a safe laboratory environment what could previously be tested only in larger, more dangerous experiments with bombs.
“In our case, it’s the laser depositing energy into a target, but you get the same formation and time-dependent evolution of uranium plasma,” author Patrick Skrodzki said. “With these small-scale explosions in the lab, we can understand similar physics.”
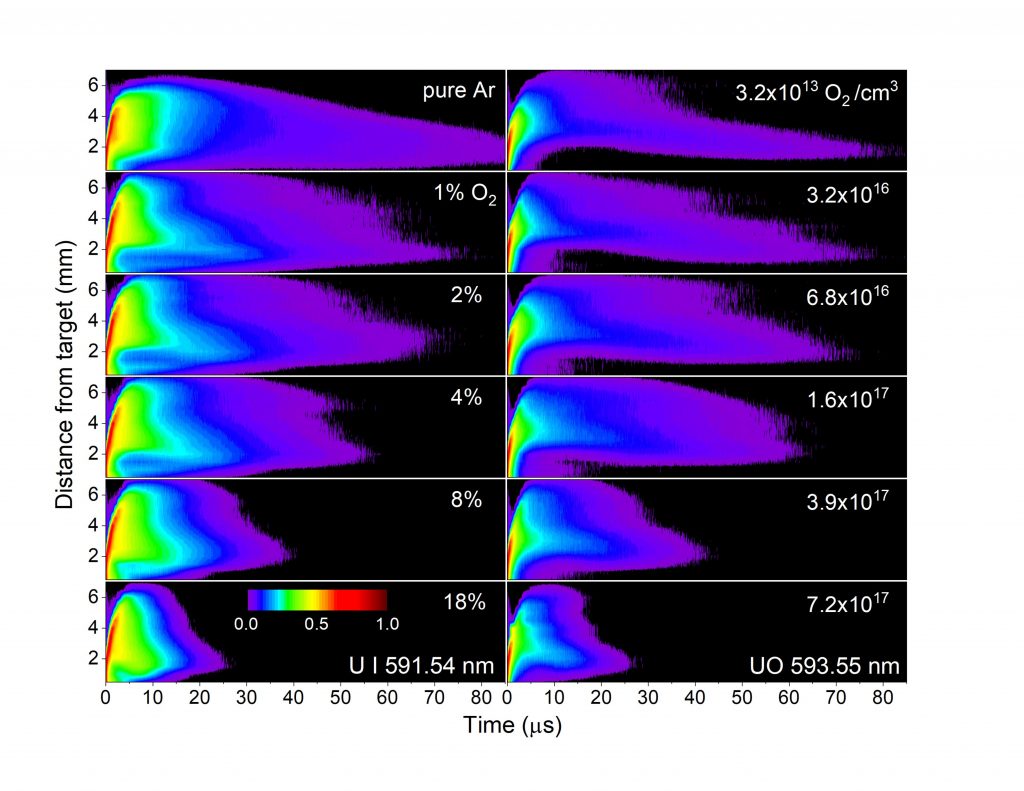
In a recent experiment, scientists working with Skrodzki used a laser to ablate atomic uranium, stealing its electrons until it ionized and turned to plasma, all while recording chemical reactions as the plasma cooled, oxidized and formed species of more complex uranium. Their work puts uranium species and the reaction pathways between them onto a map of space and time to discover how many nanoseconds they take to form and at which part of the plasma’s evolution.
In their paper, released this week in Physics of Plasmas, from AIP Publishing, the authors discovered uranium forms more complex molecules, such as uranium monoxide, uranium dioxide and other, larger combinations, as it mixes with different percentages of oxygen.
“We used optical emission and looked at excited states decaying into ground states, but that’s only a small fraction of the picture,” Skrodzki said.
Uranium, with its 92 electrons and approximately 1,600 energy levels, can produce a complicated spectrum that is hard to decipher, even with high-resolution spectroscopy. In the paper, the authors focused on one energy transition in the plasma. They closely examined the morphology of the plasma plume, collisional interactions with various concentrations of oxygen, and other factors, like plume confinement and particle velocities, to create a detailed picture of species evolution from atomic uranium to more complex uranium oxides.
The resulting data has implications for technologies that use lasers to probe materials and detail their elemental composition, such as the laser spectroscopy system on the Mars Curiosity rover. It can also be used for a portable device for verifying nuclear treaty compliance by testing for evidence of enriched uranium production.
“There’s still so much work left to do on this topic,” Skrodzki said. “It’s a scientific question, because nobody knows anything about the optical emission in the visible region from those higher oxides. We want to try to provide data to fill in those gaps.”
###
For more information:
Larry Frum
media@aip.org
301-209-3090
Article Title
Plume dynamics and gas-phase molecular formation in transient laser-produced uranium plasmas
Authors
P.J. Skrodzki, M. Burger, I. Jovanovic, M.C. Phillips, J. Yeak, B.E. Brumfield and S.S. Harilal
Author Affiliations
University of Michigan, Pacific Northwest National Laboratory, Opticslah LLC
