From the Journal: APL Materials
WASHINGTON, February 2, 2021 — Some promising biosensors and medical devices work well within pristine laboratory environments. However, they tend to stop working to deliver medical therapeutics or monitor chronic health issues once exposed to the real-world conditions of complex biological fluids.
A thick layer of foulants will quickly cover biosensors, and there is no good way to revive them once they quit working. Essentially, a biosensor is only as good as its antifouling properties.
In APL Materials, from AIP Publishing, Aleksandr Noy and Xi Chen, of Lawrence Livermore National Laboratory, review a variety of approaches developed to combat fouling. These approaches encompass physical barriers, chemical treatments, nonstick surfaces, and selective membranelike coatings that form “gates” to only allow certain species to reach a sensor’s working surface.
“There is a whole universe of very clever and quite effective approaches to protect biosensors from fouling,” said Noy. “Researchers have their pick of the technology they can tailor to the particular type of sensor they want to design.”
But despite all of this progress, Noy and Chen point out fouling remains a stubborn problem that can still wreck a good biosensor.
“Further development is needed to increase our arsenal of robust antifouling protection methods,” Noy said.
Fouling occurs in a four-stage process. First, surfaces immediately become coated with a small layer of molecules. Second, this layer gets covered with the main layer of foulant. Third, the fouled surface begins growing biofilms. Fourth, the biofilm progresses to macrofouling, which usually occurs within days or weeks.
The goal is to suppress the initial attachment of molecules, because it is incredibly difficult to remove biofilms once they form.
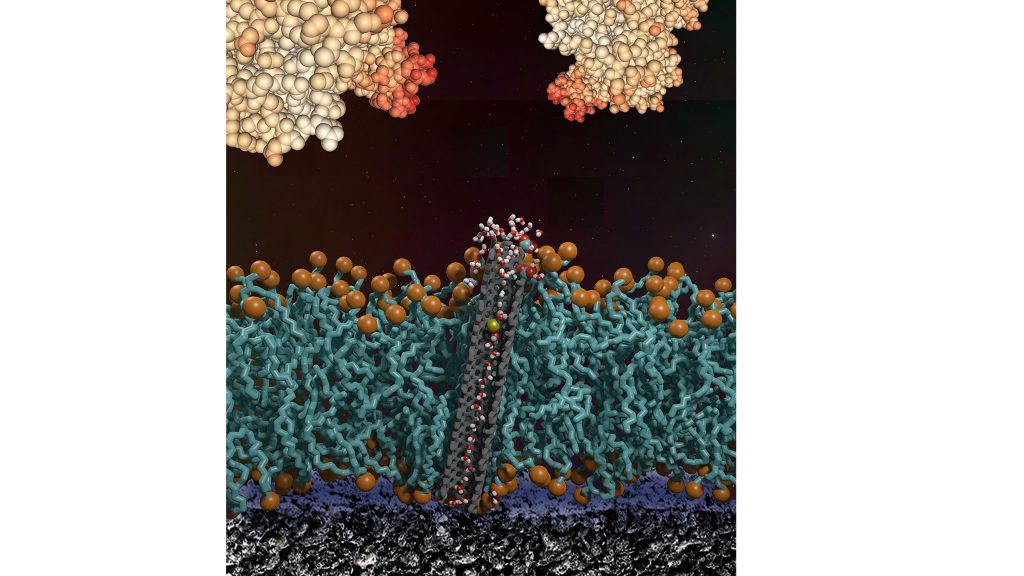
One example of antifouling protection, based on Noy’s own work, is a pH sensor with silicon nanowire transistors that are protected by a phospholipid membrane with carbon nanotube pores embedded within the membrane.
“Silicon nanowires are elegant, small, and efficient pH sensors that provide straightforward electrical signal that is modulated by solution pH,” he said. “Unfortunately, any time they come into contact with a real biological medium they foul up and cease to function.”
To get around this, his approach covers the sensors with a lipid membrane to provide a very robust protein fouling protective barrier.
“To allow protons to pass through this barrier, we embedded tiny carbon nanotube pores within the membrane,” Noy said. “These pores happen to be the most effective proton conductive channel known, so they provide an ideal conduit for shuttling protons across the protective barrier.”
Sensors protected this way “can withstand three-day exposure to protein solutions, milk, and even blood plasma and still measure pH quite well,” he said.
###
For more information:
Larry Frum
media@aip.org
301-209-3090
Article Title
Antifouling strategies for protecting biolectronic devices
Authors
Xi Chen and Aleksandr Noy
Author Affiliations
Lawrence Livermore National Laboratory
