From the Journal: Chemical Physics Reviews
WASHINGTON, May 25, 2021 — For millennia, silver has been utilized for its antimicrobial and antibacterial properties. Although its use as a disinfectant is widely known, the effects of silver’s interaction with bacteria on the silver itself are not well understood.
As antibiotic-resistant bacteria become more and more prevalent, silver has seen steep growth in its use in things like antibacterial coatings. Still, the complex chain of events that lead to the eradication of bacteria is largely taken for granted, and a better understanding of this process can provide clues on how to best apply it.
In Chemical Physics Reviews, by AIP Publishing, researchers from Italy, the United States, and Singapore studied the impacts an interaction with bacteria has on silver’s structure.
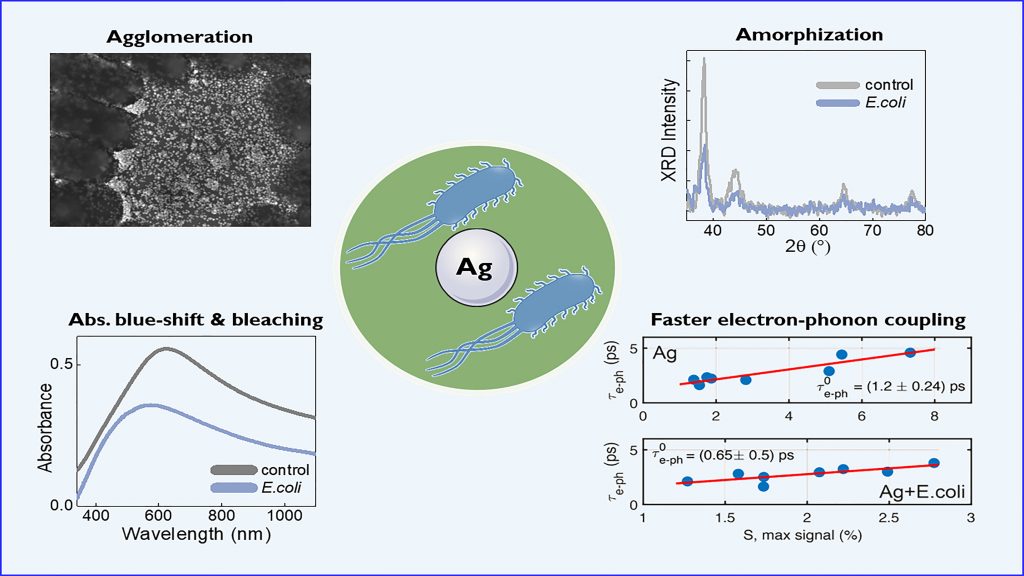
When monitoring the interaction of silver nanoparticles with a nearby E. coli culture, the researchers found the silver undergoes several dramatic changes. Most notably, the E. coli cells caused substantial transformations in the size and shape of the silver particles.
It is often assumed the silver stays unmodified in this process, but the work done by the team shows this not to be true.
The electrostatic interaction between the silver and the bacteria causes some of the silver particles to dissolve as it releases ions to penetrate the bacterial cells. This dissolution modifies the shape of the silver particles, shrinking and rounding them out from triangular shapes into circles.
These effects are even more pronounced if the E. coli cells are pretreated with a molecule to increase the permeability of their membranes before they meet the silver.
“It seems from this study that silver is ‘consumed’ from the interaction,” said Guglielmo Lanzani, one of the authors on the paper and director of the Center for Nano Science and Technology of IIT-Instituto di Tecnologia.
Fortunately, this “consumption” likely does not impact silver’s antimicrobial properties, because the effect is so small.
“We think this does not affect the efficiency of the biocidal process and, due to the tiny exchange of mass, the lifetime is essentially unlimited,” said Giuseppe Paternò, a researcher at IIT and co-author of the study. “The structural modifications, however, affect the optical properties of the metal nanostructures.”
Direct investigations of processes like these are difficult, because laboratories are controlled environments that cannot fully capture the complexities of a biological setting of bacterial cells.
Nevertheless, the group is planning further experiments to explore the chemical pathways that lead to the structural changes in silver. They hope to uncover why silver works better than other materials as an antibacterial surface, and why bacterial membranes are particularly vulnerable to silver, while other cells remain less affected.
###
For more information:
Larry Frum
media@aip.org
301-209-3090
Article Title
The impact of bacteria exposure on the plasmonic response of silver nanostructured surfaces
Authors
Giuseppe Maria Paternò, Aaron Michael Ross, Silvia M. Pietralunga, Simone Normani, Nicholas Dalla Vedova, Jakkarin Limwongyut, Gaia Bondelli, Liliana Moscardi, Guillermo C. Bazan, Francesco Scotognella, and Guglielmo Lanzani
Author Affiliations
Instituto Italiano di Tecnologia; Politecnico di Milano; Consiglio Nazionale delle Ricerche; University of California, Santa Barbara; Nanyang Technological University
